Pythium oligandrum Species of Pythium (in the cellulose-walled fungal group, Oomycota) are best known as pathogens of crop plants. They cause damping-off and other seedling diseases, and also progressively destroy the root tips of older plants, leading to 'decline' diseases of orchard trees and other perennial crops. However, a few Pythium species are aggressive parasites of other fungi, termed mycoparasites. Pythium oligandrum is an example of this. It has an astonishingly fast mode of action and can be used as a biological control agent of several plant-pathogenic fungi. Two commercial formulations are currently available for this purpose. |
|||||||||||||||||||||||||||||||
 |
|||||||||||||||||||||||||||||||
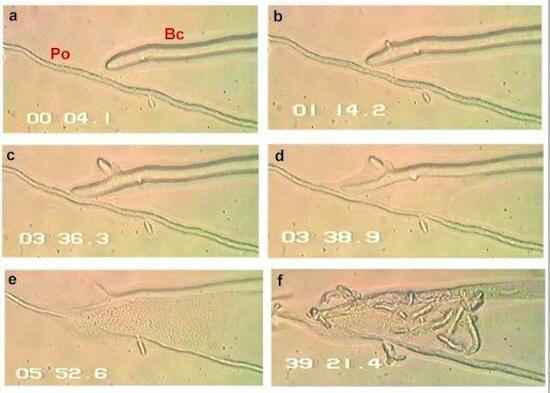
Above is a sequence of six frames (a-e) from a videotaped interaction between Pythium oligandrum (Po) and the plant-pathogenic fungus Botrytis cinerea (Bc) on a thin film of water agar. Times shown as min/sec/tenths of sec; recording started at zero time (a). The hyphal tip of Bc contacted the hypha of Po after 1 min,14 sec (b) and continued to grow for a while (c). A few minutes later (d, e) the tip of Bc suddenly lysed, expelling the protoplasm. After another 30 min (e) Po had produced many hyphal branches that grew into the spilled protoplasm of the other fungus, using the spilled contents as a nutrient source. From: Laing SAK & Deacon JW (1991) Video microscopical comparison of mycoparasitism by Pythium oligandrum, P. nunn and an unnamed Pythium species. Mycological Research 95, 469-479. |
|||||||||||||||||||||||||||||||
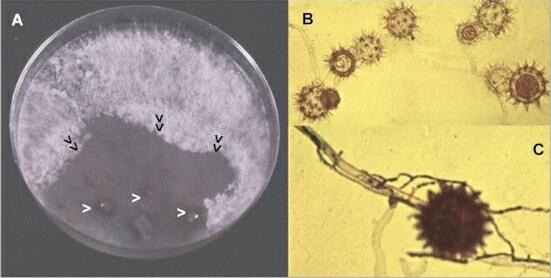
Occurrence and isolation of mycoparasites Mycoparasites are extremely common in soil, where they probably act as natural regulators of the populations of other fungi. One of the simplest ways of detecting them is to place crumbs of soil on an agar plate previously colonised by another fungus. This excludes the growth of most fungi, but enables mycoparasites to grow and sporulate. Below (A) is a plate of potato-dextrose agar precolonised by a species of Phialophora - a suitable fungus for detecting P. oligandrum in soil. Three small soil crumbs (arrowheads) were placed on the Phialophora colony, and P. oligandrum grew from the crumbs, reaching the point marked (double arrowheads) and collapsing the white aerial hyphae of the host fungus. When viewed microscopically after 5 days (B) the colonised region contained many spiny-walled oogonia (female sexual structures) characteristic of P. oligandrum. Thus the precolonised plate technique is both an isolation method and a detection method - we can record presence or absence of the fungus without needing to isolate it in pure culture. As shown in C, P. oligandrum (the narrower hyphae, stained with trypan blue) parasitises the host fungus (broader, empty hyphae) on the precolonised agar. |
|||||||||||||||||||||||||||||||
 |
|||||||||||||||||||||||||||||||
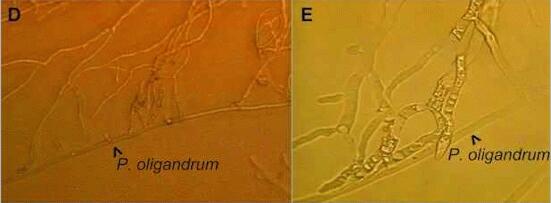
Figures D and E (below) show the remarkable effect of even a single hypha of P.oligandrum on the advance of hyphae of Fusarium culmorum across an agar plate. F. culmorum stopped on contact with the mycoparasite (D), and at higher magnification (E) its hyphae are seen to have been disrupted - the contents are highly vacuolated and coagulated.
|
|||||||||||||||||||||||||||||||
Several potential mycoparasites can grow on Phialophora plates, but different host fungi seem to select for different mycoparasites. The table below summarizes a study in which 28 different soils were plated onto three host fungi. Four mycoparasites were detected, and from the final column we see that all of them were common - they were present in at least 18 of the 28 soils sampled. However, all the mycoparasites except for Gliocladium roseum showed a strong host preference. Perhaps this type of work could help us to select particular mycoparasites for control of different plant-pathogenic fungi. Some of the more host-specialised mycoparasites such as Verticillium biguttatum (on Rhizoctonia solani) can be isolated selectively from soil by using the appropriate host-precolonised plates. |
|||||||||||||||||||||||||||||||
From: DFC Mulligan & JW Deacon, 1992. Mycological Research 96, 605-608. |
|||||||||||||||||||||||||||||||
A mycoparasite unique to Britain? No, that's hardly likely! But there is a mycoparasite reported only from Britain and described as a new species, Pythium mycoparasiticum (JW Deacon, SAK Laing & LA Berry, 1991, Mycotaxon 42, 1-8). We have found it in about 20% of all the British soils examined, by plating the soil on Phialophora plates or on other susceptible host fungi such as Fusarium culmorum or Botrytis cinerea. However, it is found in the same types of soil as P. oligandrum and it is relatively slow-growing. So the soils need to be diluted with sand before plating, to help reduce competition from P. oligandrum on the precolonised plates. This illustrates an
important point: there
are many fungi and other microorganisms remaining to be
discovered and described. Some scientists estimate that
we know only 5% of all the fungi and 1% of all the
bacterial species on earth. But, in order to find them
and to evaluate their biotechnological potential, we need
to look in unusual habitats or (perhaps more important)
to devise new methods for detecting them. |
|||||||||||||||||||||||||||||||
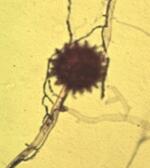
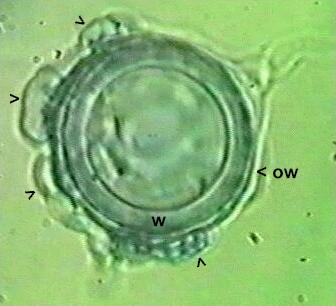 Pythium mycoparasiticum This species has oogonia with a smooth wall (ow), unlike the spiny walled oogonia of P. oligandrum. The oogonia also are enveloped by several male sex organs (antheridia, marked by arrowheads). Fertilisation has occurred in this oogonium, leading to the development of a resting spore (oospore) with a thick wall (marked w). |
|||||||||||||||||||||||||||||||
Further reading: JM Whipps (1997) Developments in the biological control of soil-borne plant pathogens. Advances in Botanical Research 26, 1-134. Website:
|
|||||||||||||||||||||||||||||||