Wood is one of the most abundant energy sources on earth, but it is difficult to degrade. Only a few specialised fungi of the groups basidiomycota and ascomycota have the ability to do this. Some of them are important pathogens of trees, including Armillaria mellea which is considered here and Heterobasidion annosum which is considered in a separate Profile. Some cause structural damage to the timbers of buildings - for example, the dry-rot fungus, Serpula lacrymans (Figure A). Most, however, are saprotrophs in natural environments, with major roles in nutrient cycling. Wood-decay fungi are of increasing biotechnological interest, because their enzyme systems can detoxify pollutants and can delignify agricultural wastes, leaving cellulose as a potentially cheap commercial substrate for industrial fermentations. Here we consider:
|
||||
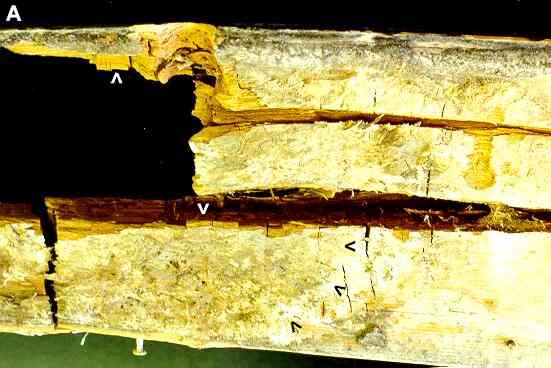
Figure A. Serpula lacrymans (dry rot). The image shows part of an internal door post with extensive dry rot. The rotten wood shows brick-like cracking (white arrowheads) typical of this fungus. At bottom left we see the sulphur-yellow spreading fans of hyphae of the fungus on the wood surface - the advancing margin is indicated by the three black arrowheads. |
||||
Special features of wood-decay fungi Wood-rotting fungi need to be specially adapted to overcome three major constraints.
|
||||
Figure B. Sapstain fungi seen in thin sections of wood stained with safranin. The low-power image (left) shows a section through two parts of the ray parenchyma traversing the xylem. The right-hand image shows darkly pigmented fungal hyphae growing in the ray parenchyma. |
||||
Types of wood decay fungi Wood-decay fungi can be grouped in various ways:
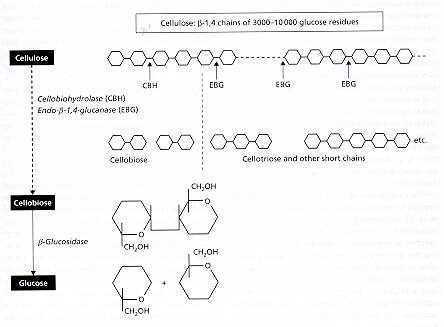
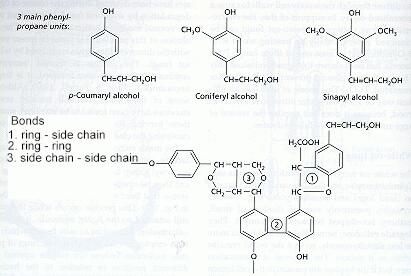
White-rot fungi These fungi degrade all the major wood components (cellulose, hemicelluloses and lignin) more or less simultaneously, so that the wood becomes progressively more fragile but remains white as it decays. White rots are caused by the two major root-rot pathogens of trees, Armillaria mellea (Figures C, D) and Heterobasidion annosum, and also by many saprotrophic fungi, including the common coloniser of stumps, Coriolus versicolor (Figure N), and the common wood-rot ascomycota Xylaria hypoxylon (Figure Q) and Xylaria polymorpha (Figure R). Brown-rot fungi Brown-rot fungi degrade the cellulose and hemicellulose but leave the lignin more or less intact as a brown framework. Only about 6% of wood-decay fungi cause brown rots, and all these fungi are members of the basidiomycota. They include Serpula lacrymans (dry-rot fungus, Figure A) and the common birch polypore, Piptoporus betulinus (Figure H). Soft-rot fungi Soft-rot fungi degrade
only the cellulose and hemicelluloses, and typically
occur in wood of high water content and high nitrogen
content. They are most commonly found in rotting window
frames, wet floor boards and fence posts, etc., where
nitrogen is "recruited" from soil or from
atmospheric contamination. Some of these fungi are common
decomposers of cellulose in soil (e.g. Chaetomium
species) and they are the least specialised of the
wood-rot fungi. |
||||