..
Many other images can be found in SPECIAL FOCUS TOPICS and PROFILES OF FUNGI. Please respect the copyright waiver
Distorted lesions caused by Taphrina populina on poplar leaves (Populus tremuloides). The yellow colour of the lesions is caused by abundant sporulation from the naked asci, shown below. This fungus is related to the more commercially damaging fungus, Taphrina deformans, the cause of leaf curl of peach and almond trees. [© Jim Deacon]
Taphrina populina, a plant-parasitic member of the Archaeascomycota which causes yellow blister of poplar leaves (Populus spp.). The fungus grows as a mycelium in the plant leaves, causing leaf distortion, and it produces naked asci which project from the surface of the leaves. Within the asci the ascospores often bud, so that the asci become filled with a mixture of ascospores and budding yeast cells (particularly evident in the top ascus). [© Jim Deacon]
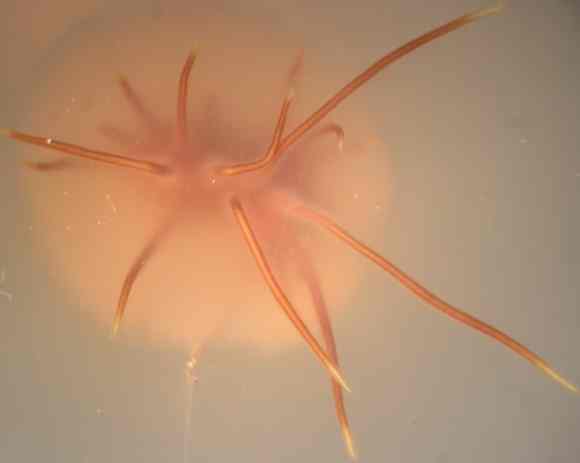
Young rhizomorphs of Armillaria, growing from the underside of a colony on an agar plate. A remarkable feature of this fungus is that it grows initially as a mycelial colony but soon produces differentiated "multicellular" rhizomorphs that grow much more rapidly than the individual hyphae. The growing tips of the rhizomorphs remain hyaline (colourless) but become pigmented behind the rhizomorph tip. The image shown is about 3 cm diameter. [© Jim Deacon]
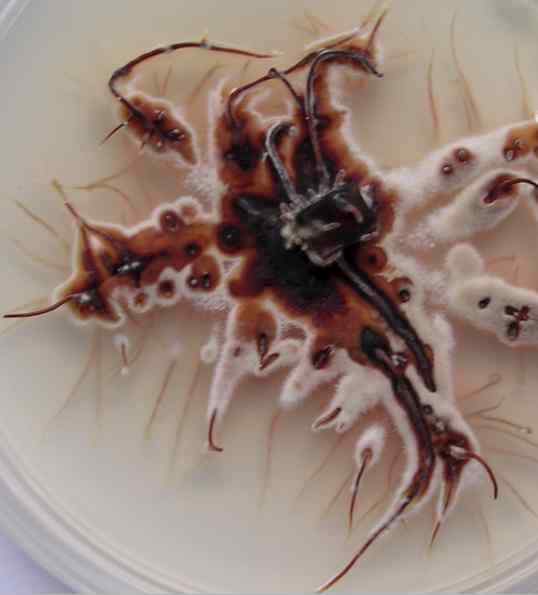
An older colony of Armillaria, viewed from the upper surface of agar in a Petri dish. Rhizomorph tips that grow into the aerated zone near the colony surface rapidly become melanised and cease growth. They then develop a fringe of hyphae along their lengths. The colony was about 7 cm diameter (20 days old) when this image was taken. [© Jim Deacon]
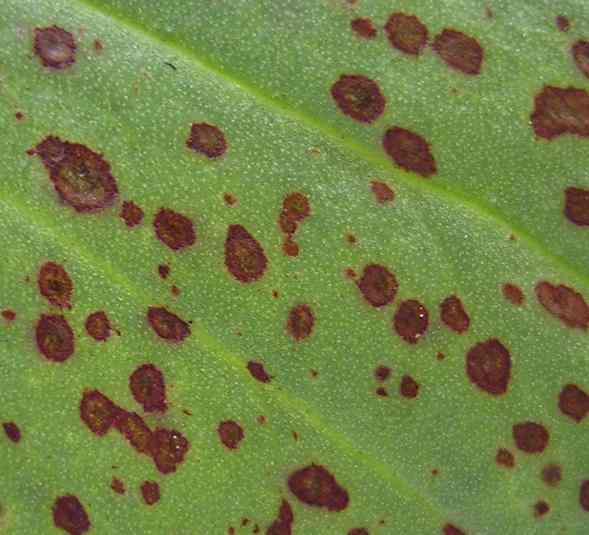
Discrete necrotic lesions caused by Botrytis fabae on the surface of a broad bean (Vicia faba) leaf. Each lesion is the result of infection from one or a few spores. The extent of disease is limited by the rapid host response to infection, involving necrosis (death) of the surrounding tissues and the concomitant accumulation of fungitoxic compounds. [© Jim Deacon]
Spreading, coalescing lesions of Botrytis fabae on a broad bean leaf. In suitable conditions that favour infection the whole crop can be rapidly destroyed. [© Jim Deacon]
Tar spot of sycamore (Acer pseudoplatanus) caused by the fungus Rhytisma acerinum (Ascomycota). This disease is one of the most common and conspicuous. The lesions start to appear in June or July, after the needle-like ascospores are released from apothecia that have overwintered on fallen leaves. The ascospores are shot from the apothecia to a height of about 1 cm but can then be carried upwards by wind to infect most of the leaves on a tree. The tar spot lesions are initially yellow but later develop a black shiny appearance. [© Jim Deacon] The incidence of tar spot is strongly influenced by atmospheric pollution, especially sulphur dioxide levels in the atmosphere. The disease is virtually absent from from highly polluted regions, but can be intense in pollution-free environments, where it can lead to severe defoliation. Sycamore is native to central and southern Europe but was introduced into Britain in the 15th or 16th Century and is now very common. It is an attractive, fast-growing tree, but it can out-compete the slower-growing native trees such as beech and oak.
Fig 12. Ganoderma lucidum fruitbodies growing from a block of compressed sawdust and woodchips. This fungus is rare in Britain, but relatively common in tropical and subtropical regions, where it can cause a serious root-rot of pine and other coniferous trees. The fruitbodies are richly laquered, with shiny black stipes. They often emerge from just below soil level at the base of infected trees. [© Jim Deacon]
Stunting and deformation of the shoots of grapevines, caused by Eutypa armeniacae (Ascomycota). This fungus can cause serious damage to grapevines and apricots, through natural wounds or pruning wounds. The fungus discharges its spores as octads (clusters of 8 ascospores) held together by mucilage, to increase the efficiency of impaction onto shoots at relatively low wind speeds (see Chapter 10). After impaction, the fungus relies on secondary spread by rain or irrigation water, which disperses the mucilage and carries the individual spores down the plant to wound sites. The shoots in the foreground are severely stunted in comparison to the healthy shoots. [© Jim Deacon] |